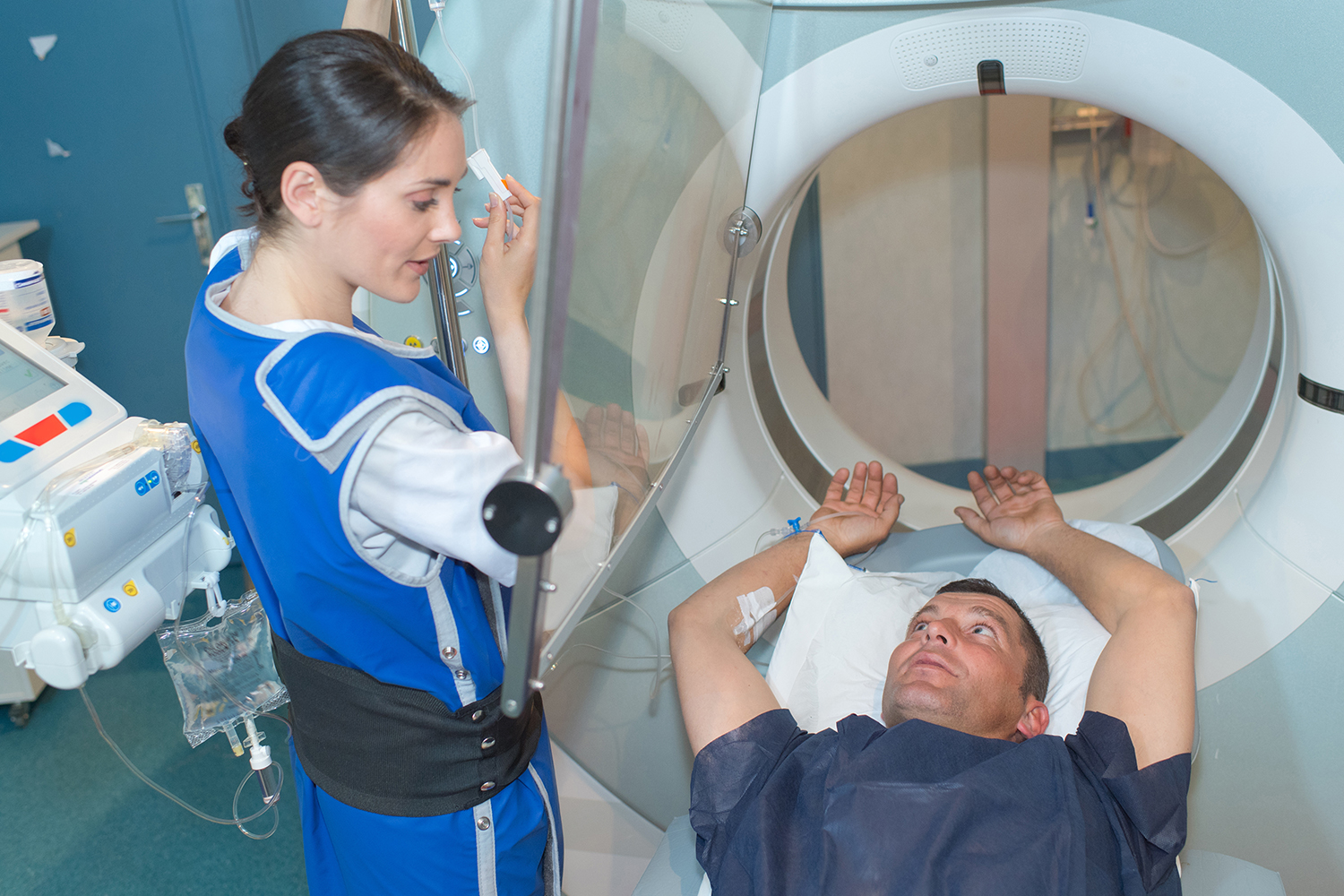
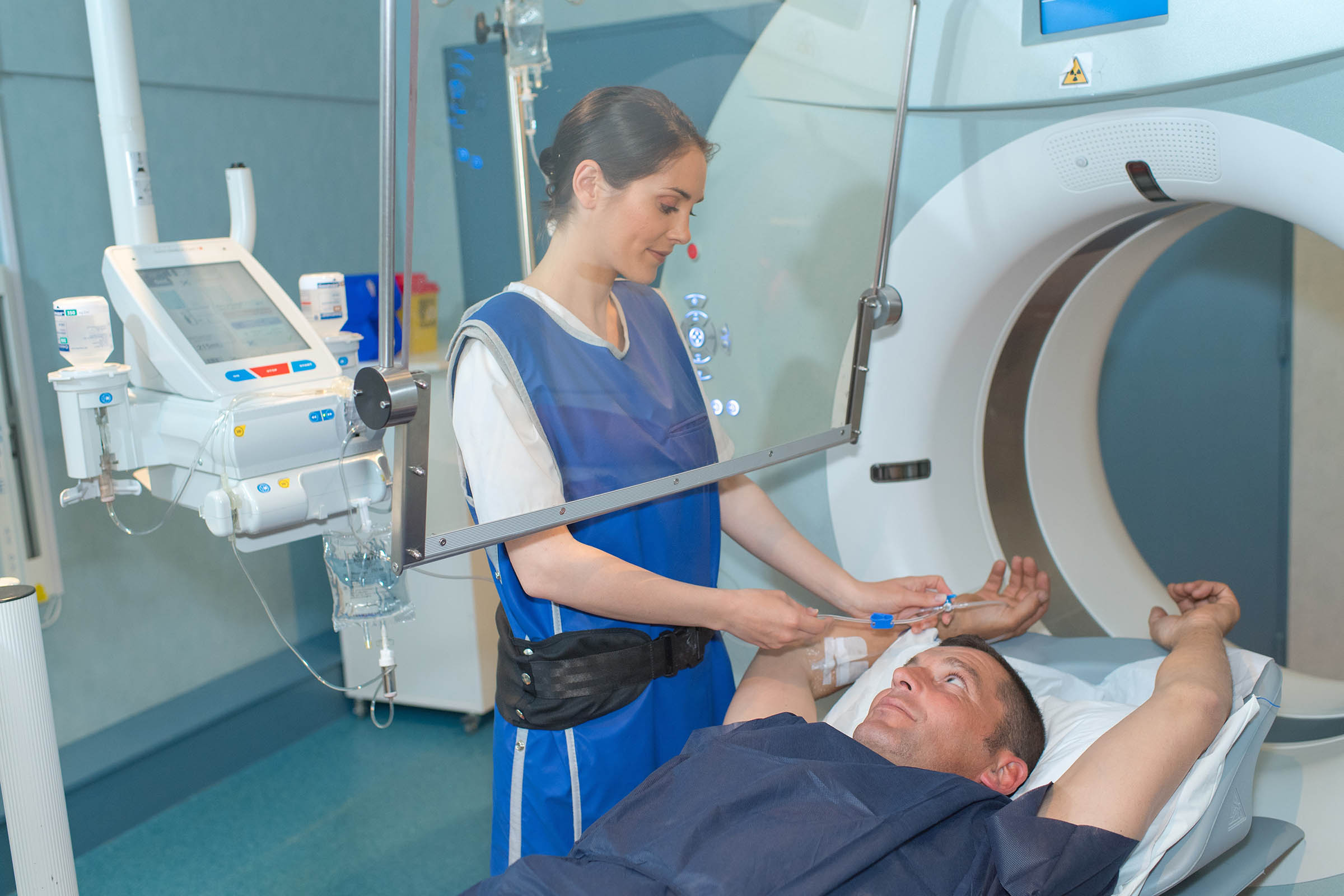
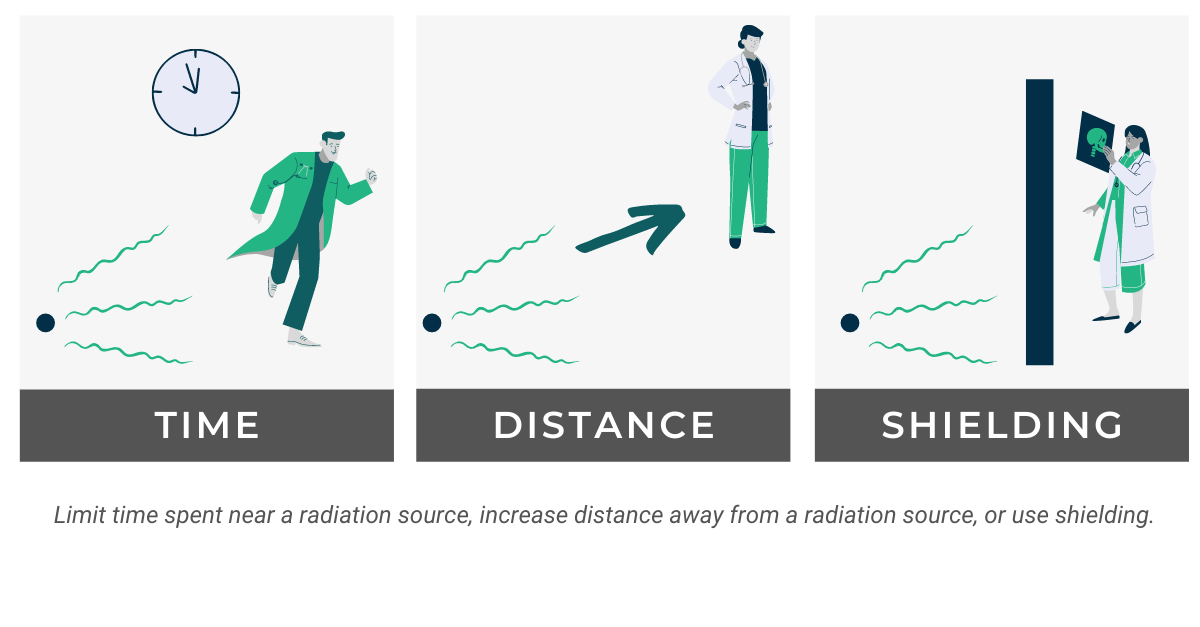
Basic radiation protection guidelines can be summed up in three simple concepts: time, distance, and shielding. While both limiting the time spent and increasing the proximity to an ionizing radiation source is something that lies within the power of the individual, shielding and X-ray room design require careful planning and execution by the facility or Radiation Safety Officer.
What is radiation shielding?
Radiation shielding is simply a barrier placed between a source of radiation and the area or person that needs to be protected. The purpose of radiation shielding is to limit, control, or modify the radiation exposure rate at a set point.
Shielding is based on attenuation or the gradual reduction in the intensity of energy through a specific medium. X-ray radiation that passes through certain materials decrease and are absorbed, thereby reducing the exposure to the other side of the barrier.
Without shielding, the public, radiation workers (including dentists and veterinarians), and even nearby office workers could be exposed to levels of radiation outside regulated exposure limits, which can potentially lead to negative health effects. Although it is impossible to completely avoid exposure to radiation, shielding is a critical consideration in any medical facility that greatly reduces unnecessary exposure.
Shielding Materials
There are several different materials that provide protection from penetrating radiation. Concrete, water, special plastic shields, air stops, and lead are all barriers that stop different types of rays and particles, reducing the overall dose a person receives.
In medical environments, the most common shielding materials used include lead, lead-free shielding, and lead composites.
Lead
Lead is one of the most used and most effective shielding materials. It is a highly dense material with a high atomic number and a high number of electrons which make it ideal for shielding in most medical radiation environments. This is because the type and energy of radiation in a medical environment that passes through lead are absorbed or scattered by the electrons present in the material.

Lead is also cheap and easy to process. It can be mixed with other materials like glass, or binders like vinyl, which allows it to be used as construction materials in X-ray rooms or worn as shielding garments.
Lead-Free Shielding
Technological advances have allowed for the creation of non-toxic, lead-free shielding materials as well. Other attenuating materials such as antimony (Sb), tungsten (W), and tin (Sn) are used in place of lead and combined with additives and binders to create wearable protective garments or materials. They offer equal protection from scatter radiation.
Lead-free shielding has several benefits, including being both recyclable and non-toxic. Lead-free shielding materials can also be lighter which makes them easier for personnel to wear during longer procedures.
Lead Composite
Lead composite shielding is a long-lasting mixture of lead and lighter materials that attenuate radiation just as successfully as traditional lead shielding barriers.
Because of lead’s weight, it can be cumbersome to use and wear for long periods of time, limiting the efficacy of a radiation worker. Lead composites solve this problem. They are made with blends of tin, vinyl, and rubber and create a shielding barrier that can be up to 25% lighter than traditional shielding without sacrificing their ability to block penetrating radiation.
Shielding and Scatter Radiation
In some diagnostic X-ray procedures, medical personnel such as operators, radiologists, and technologists are required to remain in the room with the patient. This proximity frequently exposes them to something called scatter radiation or radiation that bounces off a patient’s body during a procedure.
To limit this exposure, some medical personnel are required to wear frontal or full wrap-around style lead aprons, thyroid shields, and lead glasses/gloves. These protective garments can attenuate roughly 93% of photons at typically scattered energies.

Shielding Products and Design
There are several different ways radiation shielding can be applied or designed to protect healthcare workers.
Room Shielding
Shielding may be required in the floor, ceiling, doors, or any wall of any X-ray or radioactive material use room. Shielding is used to protect workers, patients, or the public that may be in the adjacent areas/rooms.
During a room’s construction, special shielding materials are installed where their need has been determined. These materials can include lead-lined windows and doors, lead-lined drywall or plywood, lead sheets for floors and ceilings, pipe shielding, and more.
X-ray room shielding requirements vary from state to state. It is important to consult with a qualified expert familiar with these regulations as well as work with an architect experienced in constructing X-ray suites before building a new room.
Leaded Glass and Curtains
In some cases, it isn’t possible for a facility to build shielding into the physical structure of a building.
Leaded glass barriers are a barrier used by techs and doctors which allow them to safely view a patient during an imaging procedure. This type of glass is ideal for radiation-producing equipment in the 80-300 kV range thanks to its high lead content.
Lead curtains are also used to shield radiation workers, particularly in large animal hospitals or operating rooms. These curtains are leaded rubber or vinyl sheets that are ideal for protection against low-level or secondary radiation. They make for room-saving partitions that can be open or closed as needed and typically offer protection from 0.5mm to 2.00mm lead equivalency.
Mobile Shielding Barriers
In some cases, additional barriers are needed to protect doctors and techs during radiology, nuclear medicine, cath lab, or diagnostic imaging procedure. These barriers are lead-lined partitions on wheels, often with a protected window to allow for patient observation.
Mobile radiation barriers come in a variety of shapes, sizes, and lead equivalencies. They are ideal for maintaining flexibility and ease of movement in a procedure room while successfully minimizing the scattered radiation dose to workers in the room.
Versant Physics Shielding Services
Understanding the detailed shielding requirements for your state or facility can be a time-consuming challenge. If executed incorrectly, there can be serious consequences to the health and safety of radiation workers, patients, and building staff as well as potential regulatory compliance fines.
That’s why it is important to have a radiation safety consultant like Versant Physics on your side. Whether you’re constructing a new X-ray room, remodeling or repairing an existing shielding setup, or looking to upgrade your current shielding equipment, our team of expert health and medical physicists can assist.
We provide radiation shielding calculations, evaluation, and design for facilities of all kinds, including hospitals, clinics, dentist offices, chiropractor offices, and veterinary clinics. Our range of expertise includes:
- Radiography
- Fluoroscopy
- Computed Tomography (CT)
- Nuclear Medicine/PET
- Mammography
- Dental/Veterinary X-ray
Not sure what materials or type of shielding is right for your facility? Contact our regulatory experts for a free 30-minute consultation.